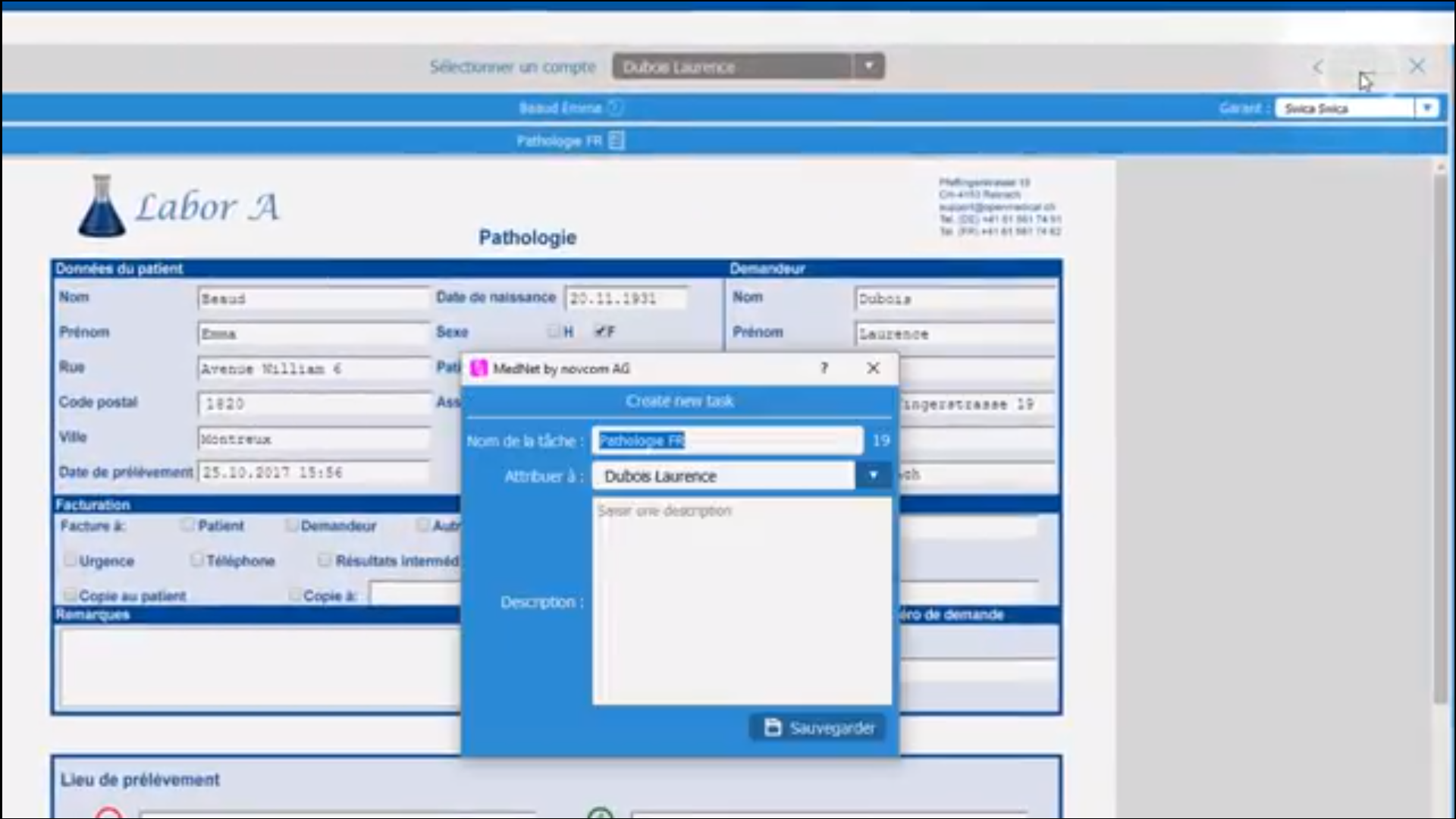
iMednet
About iMednet
iMednet Pricing
Please contact Mednet directly for pricing information.
Starting price:
$1,800.00
Free trial:
Not Available
Free version:
Not Available
Most Helpful Reviews for iMednet
2 Reviews
Anonymous
1,001-5,000 employees
Used daily for less than 2 years
OVERALL RATING:
5
EASE OF USE
4
CUSTOMER SUPPORT
3
FUNCTIONALITY
4
Reviewed October 2018
iMedNet EDC
Overall, good EDC software and I would use it for all my clinical trials.
PROSI use this for one of my clinical trials. It's not difficult to use and the functions are all there for EDC. Tech support is ok, but not excellent. It's easy on the eyes (very important because you're inputting in a lot of data and it's easy to overlook things and make mistakes, because of all the distracting sidebars, etc).
CONSTraining might take a bit, customer support can be better, and takes a while to learn where all the functions are located.
Susan
Research, 1 employee
Used weekly for less than 12 months
OVERALL RATING:
5
EASE OF USE
4
VALUE FOR MONEY
3
CUSTOMER SUPPORT
3
FUNCTIONALITY
5
Reviewed October 2017
Been using iMedNet EDC for a year but have been familiar with it for several years.
Good system with lots of functionality. Provides a tool that is easy to use and has modules for almost all activities related to clinical trials that use data, from EDC to CTMS. Provides data in formats ready for analysis.
PROSThe functionality and additional modules that allow for the data to flow easily between. The tool offers every option you might want from plain EDC to randmoization and inventory management, to ePRO, event adjudication and CTMS. You can customize the modules you need for each study, saving money.
CONSSometimes it is difficult to only get the modules you want and need for the right price. The company says that anyone can program the system, but that is not necessarily true for more complex studies. It is important to have experience in setting up the system to do it efficiently and effectively. A library of forms is not available yet. Cannot use dynamic forms based on a response for a field. Queries cannot be "printed" to send to the sites or others for resolution (i.e. a paper process). There are other small issues that I know MedNet will be working to add moving forward.
